Imagine a world where your phone can hold a charge for days, your electric car can get you across 25 percent of America on a single charge, and that taco delivery drone can finally make it out to your area. In fact, every battery-powered device you own lasts twice as long on a single charge, can recharge several times as fast, can go through five times as many charge cycles before wearing down, works in most temperatures found on Earth, and is made with environmentally-friendly, non-combustible materials.
Congratulations, you’ve just imagined a world where solid-state batteries exist and, spoiler alert: it’s here! Solid-state batteries, so called because they use a solid electrolyte (as opposed to a liquid) between the anode and cathode, are still mostly in the R&D phase, but by 2030 they’ll probably be making our phones, cars, and taco drones longer-lasting and lighter.
What’s wrong with Lithium-ion batteries?
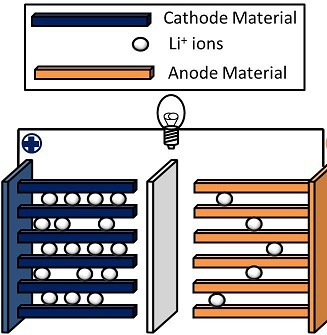
To understand what makes a solid-state battery special, you first have to know how the Lithium-ion (Li-ion) batteries that currently power most of our devices work.
There are four important components in a Li-ion battery:
- The anode (negative), where current flows in (usually made of graphite/carbon)
- The electrolyte layer, which transports ions between the anode and cathode (made of some form of liquid lithium salt – “battery acid”)
- A separator layer between the anode and the cathode (with electrolyte solution on both sides)
- The cathode (positive), where the current flows out (usually made of nickel, cobalt, manganese, and lithium)
When the battery charges, ions move through the electrolyte layer from the anode to the cathode. When it discharges, the ions move back to the anode. The exact mechanisms behind this have been steadily improving since Li-ion batteries were invented, but the fact remains that the liquid electrolyte is flammable and can combust, requiring the batteries to be less dense and have extra safety measures built in. The technology around this is improving, but there’s an upper limit to how energy-dense a cell with a liquid electrolyte can be, and at some point, we’ll probably hit it.
Why solid state is better
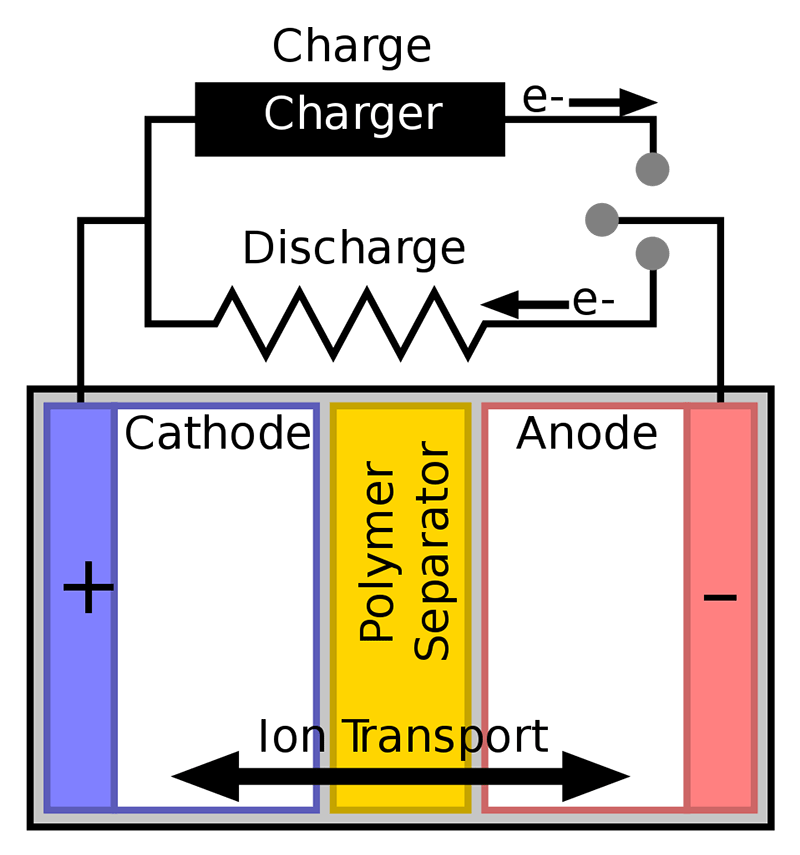
Solid-state batteries replace the liquid electrolyte with a solid one which allows the anode to be made of lithium and increases energy density by packing ions much closer than they can be in liquid. Solid materials are much more stable and can’t leak, which eliminates the need for a lot of extra safety measures. This extra stability also means they can be charged much faster and output more energy in a single burst without exploding or melting.
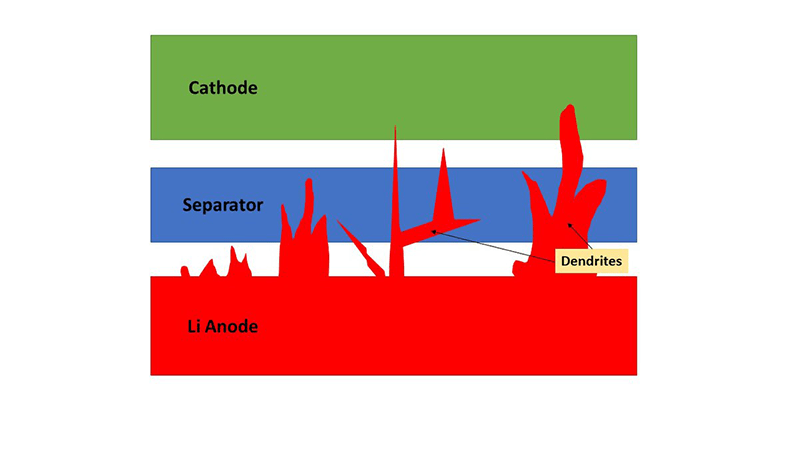
The big problem that needs to be solved for solid state to work, though, is dendrites – tiny spikes of lithium that build up in batteries as they’re cycled, damaging the structure. Dendrites cause batteries with both liquid and solid electrolytes to degrade, but it’s historically been a worse problem for solid state.
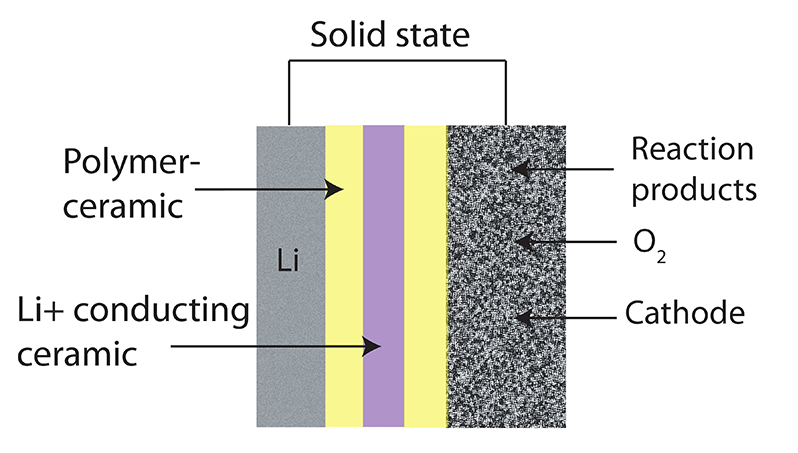
There’s a lot of brainpower and money going into cracking this issue, though, including from one of the inventors of the original Li-ion battery, John B. Goodenough. In 2017, he and his team announced that they had solved the dendrite issue and had come up with a battery using a glass electrolyte that checked pretty much all of the solid-state battery boxes.
It’s not just the one team, though: dozens of car companies, universities, and other institutions are racing to develop a viable solid-state battery, and several of them claim to be quite close. It remains to be seen which combination of materials and engineering will produce the most viable solid-state battery, but there are so many things being tried that at this point it’s essentially just a matter of waiting to see which ones work best.
Solid-state IoT
A lot of research teams are predicting that their products will be commercially viable between 2023 and 2025, but these batteries are likely to remain expensive and hard to find for quite a while. Once they manage to hit a scale and price that competes with Li-ion, though, the fact that they last longer and need less maintenance will be a big push forward for the IoT, not to mention that IoT manufacturers are already finding ways to improve battery life now or even forgo batteries altogether.

The greater energy density and throughput they can provide will make electric vehicles lighter, more powerful, and longer-range, meaning electric boats and even planes could be on the horizon alongside even more efficient electric cars.
Industrial IoT, with the huge numbers of sensors and autonomous machines it requires, will also get a lot more efficient. Replacing batteries doesn’t sound like that big of a job until you have millions of batteries to replace, at which point even small improvements could mean huge savings.

Wearables such as smartwatches, rings, and fitness trackers would also benefit quite a bit from solid-state batteries, mostly by reducing how often you need to charge them. The fact that your wearables often die in under 24 hours limits their usefulness quite a bit, and battery bulk is definitely a key issue holding back smart glasses.
Internet of Batteries
The number of IoT devices has been skyrocketing even without solid-state batteries, and once they arrivem we can expect a big jump. Cutting down on bulk while simultaneously increasing the time devices can run between charges (and dramatically decreasing charge time) isn’t an incremental step forward – it’s pretty much a paradigm shift.
Engineers spend a lot of time trying to optimize space and battery life, and they probably still will be after the solid-state battery comes along, but the shift from Li-ion to solid-state will be like going from dial-up to broadband in terms of weight, power, and charge time.
Image credits: General discharging Li battery diagram, Li-air-solid-state, Battery with polymer separator, File schematic of a Li-ion battery, Lithium dendrite