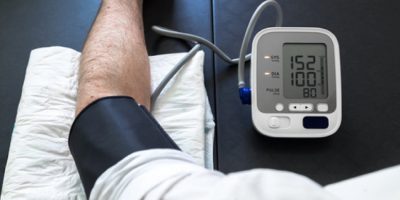
North Carolina-based Valencell is said to be a pioneer in PPG sensors that effortlessly integrate into any wearable and hearable device. Providing medical-grade accuracy in vital signs monitoring, these PPG sensors measure the biometric heart rate, heart rate variability (HRV), blood oxygenation, and now, blood pressure.
We spoke to Ryan Kraudel, vice president of marketing at Valencell, about their innovation in PPG sensor technologies and its widespread applications in the wearables and hearables market.
How Valencell’s Photoplethysmography (PPG) Sensors Work
Photoplethysmography (PPG) is a technology widely used in hospital settings to monitor vital signs, such as blood pressure, heart rate variability (HRV), and respiration rate. It uses a source of light reflecting against human tissues to detect blood volume changes and measure vital signs.
What’s “new” is transferring the same PPG technology to wearable tech and making it as accurate as electrocardiogram-based (ECG) vital sign monitors. With over 14 years of experience in developing biometric sensor technologies, Valencell is one of the first in the wearable heart rate sensors market. Their invention is a game-changer for IoT companies designing smartwatches, fitness bands and smart hearables, since they can now effortlessly integrate accurate vitals monitoring into their products.

Says Ryan: “If you’ve seen the back of a smartwatch or a fitness band with their green blinking lights, chances are it would have Valencell’s technology or some form of Valencell innovation behind it.” This is actually a credible claim, as Valencell’s unique technology is integrated in over 65 consumer wearable products, including Samsung, LG, Sony, Bose, Jabra, and Suunto.
Ryan further comments on the effortlessness with which its innovation integrates across today’s wearable technologies: “Our innovation works on the methodology of putting a shining light on the body, measuring how much of that light is reflected back, and making it ‘motion-tolerant.’ ” Evidently, once you’re able to tweak the light waveform in such a way so that it does not scatter and is no longer affected by the human body’s motion, you can build “Wearable” devices in the next step.
Are Today’s Wearable Technologies as Effective as Medical Grade Devices?
We asked Ryan if the addition of PPG sensors can bridge the difference between consumer wearables and medical grade products – for example, in blood pressure monitoring. He says it depends on “what you’re trying to measure,” “how often you’ll be measuring,” the device where the sensor is embedded, and most importantly, “where specifically on the body it is measuring the biometric signals.”
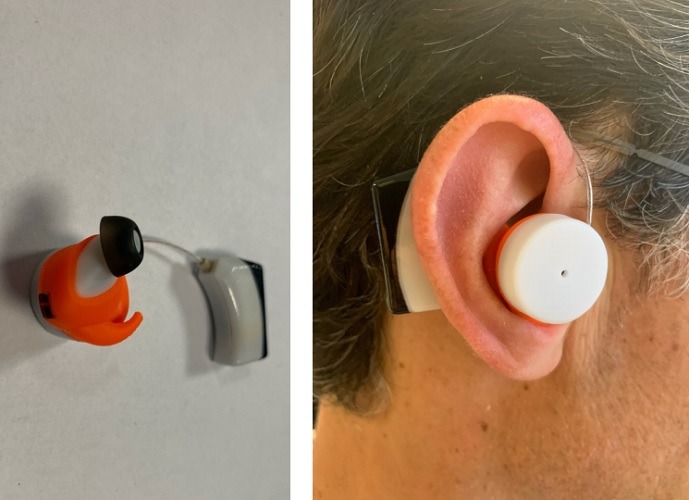
It turns out the ear is one of the best places in the body to measure biometric signals in general because it has very little local motion relative to the whole body. But specifically, it does well for vital signs, like heart rate and blood pressure. With the ear-based signal monitoring, Ryan believes “you can get gold standard accuracy.” He adds: “For heart rate, you can get an in-ear accuracy which is at par with an electrocardiogram – even during high-intensity motion.”
At the same time, other body locations may not be so accurate. For example, regarding the wrist, Ryan clarifies the problem: “They mainly consist of bone, muscle, and tendons, along with other elements of human tissue that scatter the light even further. And also, there is lot of local motion, which introduces more ‘motion artifacts’ into the signal, making it that much harder to measure.”

However, the company has also announced one the world’s first medical-grade, cuffless, wrist-worn blood-pressure monitoring sensor which is “calibration-free.” This means you don’t need to benchmark it with a cuff-based blood pressure monitoring solution. Apart from sensor data, you only need to input your age, gender, height and weight. This data is then fed into a model to generate an accurate blood pressure estimate.
What Are the Applications of PPG Sensors?
There are many non-regulated consumer wearables in the market. They all claim to measure vital signs, such as blood pressure, and may even show a systolic and diastolic number. But those figures are not accurate because they haven’t been tested for a wide range of use cases, demographics, and underlying medical conditions. For a proper testing, you need more information on real-world use cases.

Valencell works with its OEM partners in the wearables and hearables industry through an end-to-end product development cycle. It starts from the prototyping stage itself, and the collaboration reaches out into the product launch phase as well. At the heart of the testing is Valencell’s full-fledged biometrics lab, where an early form of R&D is achieved. Since they have some expertise in testing these types of devices, they also test the performance on wearables that don’t imbibe Valencell’s PPG sensor technology.
Says Ryan: “We go out of our way to work with third-party research institutions to see that the results we’re seeing in our laboratory are replicable outside Valencell. We want to make sure other people can replicate these results that we’re seeing in our labs.”

There are many advanced applications of PPG sensors that accurately measure blood pressure. Telemedicine is one of them. If consumer wearable devices can achieve the same accuracy in pressure monitoring as medical grade devices, then it has real benefits for patients in need. The abnormal spikes in pressure or other vital signs can feed the data into an early warning system which should alert the physicians.
Last Word
Tiny, comfortable and non-invasive, Valencell’s PPG sensor is pursuing FDA clearance while positioning itself as a more viable alternative to traditional cuff-based pressure monitoring devices. They already work with their OEM partners who have achieved FDA clearance with Valencell’s PPG sensor technology embedded in their devices.
Get the best of IoT Tech Trends delivered right to your inbox!